Clip Art of How Aids Affects Your T Cells
- Review
- Open up Admission
- Published:
Effects of HIV infection and ART on phenotype and function of circulating monocytes, natural killer, and innate lymphoid cells
AIDS Research and Therapy volume xv, Article number:7 (2018) Cite this article
Abstract
HIV infection causes upregulation of markers of inflammation, immune activation and apoptosis of host adaptive, and innate immune cells particularly monocytes, natural killer (NK) and innate lymphoid cells (ILCs). Although antiretroviral therapy (ART) restores CD4 T-jail cell counts, the persistent abnormal activation of monocytes, NK and ILCs observed likely contributes to the incomplete recovery of T-cell effector functions. A amend understanding of the effects of HIV infection and Fine art on the phenotype and function of circulating monocytes, NK, and ILCs is required to guide development of novel therapeutic interventions to optimize immune recovery.
Groundwork
The human innate immune system is comprised of a complex network of cellular and soluble proteins that piece of work together to provide the outset-line of defense against mutual invading pathogens prior to involvement of the adaptive immune response [1,ii,three]. Innate immune cells including monocytes, natural killer cells (NK), innate lymphoid cells (ILCs), and other antigen presenting cells (APCs) play a crucial role in the ushering in the adaptive arm of the immune response [4, v]. In particular, monocytes are precursor cells to professional APCs involved in allowed surveillance [vi]. In addition, they have pattern-recognition receptors (PRRs) that find conserved pathogen-associated molecular patterns (PAMPs) which lead to the induction of inflammatory responses that combat invading pathogens [7]. Natural killer cells produce cytokines; particularly interferon-gamma (IFN-ɣ) which activates phagocytic cells and primes APCs for interleukin 2 (IL-two) secretion thus shaping adaptive immunity towards a T helper 1 (Th1) response [viii, nine]. ILCs chop-chop secrete immunoregulatory cytokines which makes them provide protective amnesty early on during infection [10] and also maintain abdominal homeostasis by directly regulating T cells through the presentation of peptide antigens on major histocompatibility complex 2 [xi].
During HIV infection, the adaptive immune system is direct affected through the rapid infection of CD4 T-cells [12] merely the furnishings on the innate immune system are more indirect through microbial translocation, inflammation, and immune activation [13]. Immune activation and inflammation cause a reduction in the numbers of monocytes, NK and ILCs, consequently leading to poor innate and adaptive allowed responses, all which result in suboptimal response to infecting antigens [14].
Antiretroviral therapy (Art) suppresses HIV replication, restores CD4 T-cell numbers, reduces microbial translocation, inflammation, and aberrant T-cell activation [15,16,17]. The net effect of this is the nearly restoration of the allowed system to pre-infection condition and command/prevention of opportunistic infections and other AIDS-associated ailments [eighteen, 19]. Several studies have still demonstrated incomplete recovery of the adaptive immune responses including ours which showed lower CD4 T-prison cell proliferation among Fine art-treated adults (with CD4 counts restored to 500 cells/µl and more), relative to their HIV-negative counterparts [19,20,21]. Innate immune cells; in particular NK cells, ILCs and monocytes, participate in the initiation and development of adaptive immune responses although little is known well-nigh their recovery during Art. This review discusses the recovery of monocytes, NK and ILCs during ART, because of their respective contributions to the regulation of the adaptive immune response.
Figure ane summarises the effects of HIV infection and ART on monocytes, NK cells, ILCs, and other innate cells. Persistent inflammation and activation of monocytes, NK cells, and ILCs is likely associated with the persistent T-prison cell activation and impaired effector functions observed amid Fine art-treated adults [20,21,22]. There is likely a unique miracle of innate immune cell recovery during Art, among residents in sub-Saharan Africa (SSA) where several endemic infections activate the immune systems. A better agreement of innate prison cell dysfunctions and their effects on the adaptive responses during Art would guide the development of innovative therapeutic intervention to optimize recovery of host immune responses.
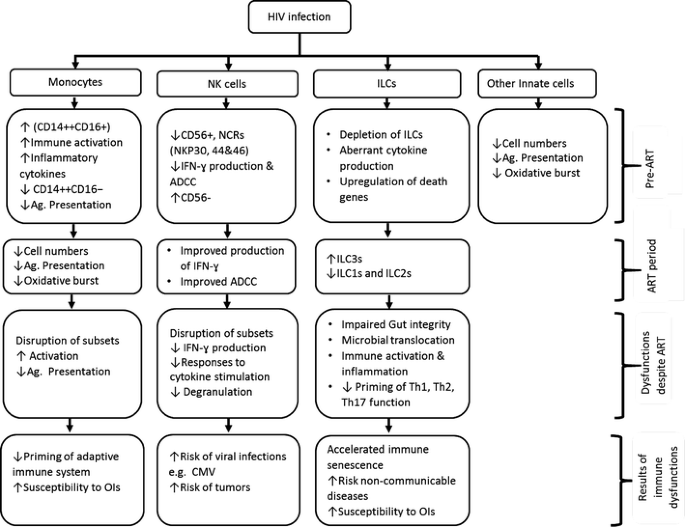
The furnishings of HIV infection and ART on monocytes, NK cells, ILCs
HIV infection and innate allowed cells
Monocytes
In the showtime few weeks of HIV infection, there is a massive aggregating of CD8 T-cells and a massive depletion of CD4 T-cells in the gut, followed by increased gut permeability and translocation of microbial products into apportionment [23, 24]. Microbial translocation contributes to increased monocyte activation as evidenced by the rapid shift in the circulating monocyte puddle from the classical phagocytic monocytes (CD14++CD16−) to the intermediate inflammatory monocyte subpopulation (CD14++CD16+) in the offset 2 weeks of HIV infection [25]. Subsequently, monocyte subsets are disrupted, leading to suboptimal effector functions of phagocytosis, intracellular killing, chemotaxis and cytokine production [26]. HIV infection, both through direct infection and indirectly through microbial translocation, leads to monocyte activation and abnormal release of pro-inflammatory cytokines including TNF-α, IL-1β and IL-6, thereby activating the immune organization [17]. In add-on to aberrant cytokine production, HIV-associated monocyte activation leads to increased release of chemokines, leading to not-specific movement of monocytes into various tissue sites [23]. Direct HIV infection of the monocytes down regulates MHCII expression, inhibits MHCII-antigen circuitous formation and reduces the monocyte power to take upward antigens for processing and presentation to T cells [27]. In a study conducted in Beijing, Chen et al. [28] demonstrated that acute HIV-1 infected individuals had significantly increased proportions of inflammatory monocyte subsets and upregulated expression of the HLA-DR and CD163 receptors when compared with HIV negative individuals. These acquired defects in monocyte function cause the inability of monocytes to present antigens [27].
We postulate that the observed increment in inflammatory monocytes and immune activation markers could further impair monocyte responsiveness to antigens making the HIV infected individuals more susceptible to opportunistic infections. After several months to years of HIV infection, viral load levels gradually increase, while CD4 T-cells proceed to reduce in number and function [29,thirty,31]. Similarly, there is dysregulation of monocyte subsets with higher populations of inflammatory (CD14+CD16+) monocytes than phagocytic (CD14+CD16−) populations. Monocytes in apportionment become functionally anergic due to continued activation and high inflammatory status [32]. Untreated, individuals with chronic HIV-1 infection go on to accept increased proportions of both the intermediate and non-classical monocytes subsets [33]. Moreover, levels of expression of CD163, a marker of activation on inflammatory monocytes remains significantly higher among individuals with chronic HIV-one-infection than HIV negative controls [34]. Protracted expression of the activated phenotype of monocyte subsets has a directly association with illness progression [33].
Art downwardly regulates the excessive production of cytokines by the inflammatory monocytes thereby reducing the levels of allowed activation and inflammation [17]. In a review by Burdo et al. [35], it was observed that Fine art initiation within the start twelvemonth of HIV infection reduced monocyte activation, as evidenced by a reduction in expression of activation marker CD163 and accented numbers of inflammatory monocytes. Similarly, markers of microbial translocation [lipopolysaccharide poly peptide (LPS), IL-6, 16S ribosomal DNA and soluble CD14], and inflammatory markers such as d-dimer and interferon-α declined with Fine art initiation [33, 36]. Although a lot of immune functions announced to be recovered during Art, some monocyte dysfunctions persist. Later on i year of therapy, ART-treated adults were reported to however have elevated levels of the inflammatory monocyte subset (CD14++CD16+) and a downregulated expression of phagocytic monocyte subset (CD14++CD16−), resulting in the reduced ability of monocytes to procedure and present antigens to T cells [34, 37]. Similarly, phagocytic activity and oxidative flare-up of neutrophils and monocytes remained impaired among HIV-i infected patients, in Athens general hospital subsequently iii months of ART [26, 38]. Yet, there is paucity of information on monocyte activation and functional recovery beyond ii years of ART, peculiarly in sub-Saharan Africa (SSA) where monocyte frequency and functional recovery has non been widely studied in HIV treatment cohorts. Given the increasing numbers of individuals receiving Fine art, for vii years and over, a better understanding of the effects of long-term ART on inflammation and monocyte activation would be relevant to inform innovations against chronic inflammation and its complications among adults living with HIV.
Natural killer cells
Natural killer (NK) cells have an important role in controlling acute HIV infection, through rapid division and production of huge amounts of IFN-γ cytokine [viii]. Strong NK cell activeness and cytotoxic receptor expression are associated with preservation of CD4 T cells and lower viral set point [39]. HIV infection is associated with several changes in the NK cell compartment, including phenotypic and functional abnormalities that contribute to difficulty in the command of HIV progression [40]. Show of dysfunctional NK cell populations has been revealed by studies in the nonhuman primate model which have demonstrated anergic NK cell accumulation in lymph nodes in SIV infection [41].
In humans, acute HIV infection generally causes activation and expansion of the whole puddle of NK cells [42], with aberrant distribution of the NK cell subsets. Pro-inflammatory NKCD56bright populations are reduced, while the cytolytic CD56dimCD16pos NK cell and dysfunctional CD56negCD16pos NK cells are increased in HIV positive people compared to HIV negative individuals [43, 44]. HIV infection reduces expression of the natural cytotoxicity receptors (NCR), NKp30, NKp44 and NKp46 [39, 45], which are essential in the containment and clearance of HIV virus. Show has farther suggested that astute HIV infection activates the upregulation of stress ligands for cytotoxicity receptors including NKG2D which leads to lysis and cell death [46].
With connected viral replication, the CD56dimCD16pos NK prison cell subset previously expanded in acute infection drib in numbers and office, as demonstrated by reduced CD107a expression and cytokine secretion [47]. The reduced numbers of cytolytic and cytokine producing NK cells would suggest that HIV infected patients with chronic illness remain susceptible to many infections peculiarly those of viral origin [42, 48]. Evidence of reduced cytokine producing NK cells was farther demonstrated in a rural Ugandan cohort, where chronically infected HIV individuals had lower expression of the NKG2A+CD57+CD56dim subset in HIV infected group than the HIV negative controls [49]. In addition to down-regulated cytokine product, HIV infection causes a reduced power of NK cells to perform ADCC due to a reduction in the number of the cytolytic CD56dimCD16+NK cells population [l] and a reduction in the intracellular stores of perforin and granzyme A [51].
Several discrepant results take been reported on the recovery of NK cells with Art.
Fine art has been shown to restore NK cell numbers with a mature phenotype in HIV-infected individuals, although defects of subset distribution and impaired ability to produce IFN-ɣ cytokine persist [44, 52, 53]. Frias et al. [54] reported incomplete recovery of NK cell subsets after 3 years of ART in Spain, in spite of undetectable viral load and an outstanding increase in the CD4 count to levels above 500 cell/µl. On the contrary, Mavilio et al. and Luo et al. [twoscore, 55] reported complete recovery of NK cell subsets and functional profiles subsequently two years of ART when compared with HIV negative individuals. In Ottawa at the immunodeficiency dispensary, NK cell cytolytic activity was greatly reduced to levels similar to those among HIV negative people, after ane twelvemonth of Art [56]. In a Ugandan accomplice, we demonstrated increased pro-inflammatory CD56bright NK cells that were associated with suboptimal immune recovery despite 4 years of suppressive therapy [57]. Given that almost of the bear witness on the recovery of NK cells is from individuals subsequently a brusque duration of ART, at that place is need to explore recovery of NK cell part after longer durations of ART. This is particularly important for sub-Saharan Africa which has more than 10 meg people receiving ART majority of whom have been on treatment for more than x years [58].
Innate lymphoid cells (ILCs)
Innate lymphoid cells are a grouping of innate immune cells that belong to the lymphoid lineage merely do non respond in an antigen-specific manner, because of their lack of a B or T cell receptor [59]. These cells are subdivided into ILC1, ILC2 and ILC3 and these mirror the CD4 T helper cells TH1, TH2, and TH17 cells in the cytokines they produce. ILCs are mainly establish at mucosal surfaces where they human action as gatekeepers to invading infectious agents, including HIV [60]. ILCs rapidly secrete immunoregulatory cytokines which makes them provide protective immunity early on during infection [61]. Studies done in non-human primates revealed that ILC populations in the gut mucosa are significantly reduced in numbers due to an increase in cytotoxicity and inflammatory cytokine product by both ILCs and NK cells during acute Simian immunodeficiency viruses (SIV) infection. The reduction in ILC numbers contributes to the massive apoptosis and dysregulation in the gut-associated lymphoid tissue (GALT) [62, 63]. Xu et al. [64] showed that IL-17 producing ILC populations were drastically reduced in acute SIV infection, especially in the jejunum.
In humans, acute HIV infection has been associated with lymphoid tissue destruction of gut mucosa and further causes upregulation of genes associated with ILCs cell decease, equally evidenced past the depletion of ILCs both in blood and gut tissues of HIV-1 infected patients [63]. The destruction of lymphoid tissue cells has been associated with microbial translocation, immune activation and disease progression in both ART-treated and untreated individuals [65]. In a study past Kløverpris et al. amid individuals with acute HIV infection in South Africa, information technology was demonstrated that all three subsets of ILCs were massively depleted from peripheral blood 7–14 days after HIV infection and these did not increment with viral load decreasing [65, 66].
In chronic HIV infection, ILC3s are further depleted and the depletion was attributed to the presence of excessive production of type 1 interferons by the plasmacytoid dendritic cells [67]. Although ILCs are depleted irreversibly from peripheral blood and the mucosal tissues, Mudd et al. [67] demonstrated that ILCs in tonsillar tissue are not significantly altered, pregnant that ILC depletion is not generalised just rather compartmentalised and with continued therapy they may exist redistributed dorsum in peripheral claret and mucosal tissues.
Initiation of Fine art during acute HIV infection, preserves ILC numbers if information technology is initiated earlier peak viremia [66]. However, ART initiation during chronic HIV disease seems to accept little event on recovery of ILC numbers; with circulating ILC1s and ILC2s remaining significantly depleted and incomplete reconstitution of circulating ILC3s even with two years of ART [65, 66]. Kramer et al. studied ILC distribution in the gut and observed that despite effective use of ART, ILCs in HIV infected individuals remain dysregulated compared to their HIV negative counterparts. This lack of recovery of ILC distribution may contribute to the loss of intestinal barrier integrity and allowed activation [66, 68, 69]. It is likely that individuals with persistent ILC dysfunction remain with limited mucosal protection and subsequently high risk of bacterial infections, autoimmune diseases and allergic infections due to the subsequent limitations in TH1, TH2 and TH17 functions that are mirrored by ILC1, ILC2 and ILC3 phenotypes, respectively [68].
Consequences and clinical implications of persistent dysfunction of innate immune cells during ART
Innate immune cells and IRIS
Fine art by and large leads to viral suppression, comeback of immune function, and improve outcomes for many HIV positive individuals. Up to 10–25% of Art-treated individuals [70, 71] may develop the immune reconstitution inflammatory syndrome (IRIS) during the kickoff months to years of HIV treatment. IRIS is a paradoxical inflammatory syndrome resulting from increased host immune responses to pre-existing opportunistic pathogens, including Mycobacterium avium complex, M. tuberculosis, Cryptococcus neoformans, Cytomegalovirus, JC virus, Pneumocystis jirovecii, Herpes zoster (VZV), and hepatitis B, as a outcome of CD4 T-cell restoration and interferon gamma product during the first months of ART [72]. The contribution of the different innate allowed cells to IRIS has been documented by dissimilar groups; for case, in a Ugandan accomplice, Tran et al. [73, 74] reported that monocyte-associated biological processes and functions were disturbed in TB-IRIS patients (after 2 weeks of Art), with dysregulation in both anti- and pro-inflammatory processes in monocytes. Andrade et al. [75] evaluated soluble biomarkers of inflammation and monocyte activation in patients who had been on ART for 12 weeks with TB-HIV co-infection from Republic of india and South Africa and establish increased plasma levels of sCD14 and sCD163 pre vs post IRIS which are strong indicators of monocyte activation and predictors of expiry in TB-IRIS patients. Natural killer cells likewise showroom phenotypic and functional differences in patients who develop IRIS relative to those who do not. At a Themba Lethu clinic Johannesburg, patients who developed IRIS had significantly college levels of NK-cell degranulation earlier Fine art initiation [76] and exhibited loftier immune activation levels as demonstrated by the elevated levels of CD69 and HLA-DR [77]. Increased NK jail cell degranulation can cause lysis of cells infected with antigens, thereby increasing the circulating antigen load in these patients and contributing to the observed IRIS [76]. Given the propagating function of monocyte activation in IRIS processes, information technology is probable that therapeutic interventions to minimise monocyte activation might indirectly modify the run a risk and severity of IRIS among ART-treated adults.
Persistent immune activation and non-AIDS complications
Aberrant activation of the innate allowed system is persistent despite Art [78], and it could exist direct due to replicating HIV virus or indirectly through co-infections including subclinical Cytomegalovirus (CMV) infection [79]. Innate immune activation can be due to HIV directly infecting the monocytes/macrophages and dendritic cells or indirectly through HIV gene products like envelop proteins of gp120 and Nef that cause activation of lymphocytes and macrophages to produce pro-inflammatory cytokines and chemokines [80]. Show of indirect allowed activation suggests that persistent leakage of lipopolysaccharide (LPS) into blood circulation [36] causes monocyte activation [81].
Persistent activation of innate immune cells is associated with the heightened product of pro-inflammatory cytokines (IL-1β, TNFα and IL-6) which cause T-cell activation. T-prison cell activation subsequently increases intracellular NF-κB levels which enhances the transcription of integrated virus and product of new virions that further infect more than cells [82]. Activation of T-cells promotes T-jail cell depletion through upregulation of apoptosis, ADCC, and by-stander killing; all of which are functions of innate allowed cells [3]. Reduced numbers of innate monocytes, NK and ILCs, consequently atomic number 82 to poor innate and adaptive immune responses causing suboptimal response to infecting antigens [fourteen]. Moreover, persistent inflammation and activation have been associated with fatal not-AIDS illnesses such as cardiovascular diseases, malignancies and organ impairment among adults crumbling with HIV [83,84,85]. The high levels of inflammation and allowed activation associated with chronic HIV illness, despite Fine art, contribute to accelerated allowed aging and increase the risk of non-AIDS illnesses including cardiovascular diseases [86, 87], cataracts [88], malignancies [89, 90], bone demineralization [91], renal affliction [92] and cognitive pass up [93]) among HIV-positive adults relative to their HIV-negative counterparts. We, therefore, postulate that strategies to downgrade innate immune cell activation and associated dysfunctions could modify the magnitude, duration, and systemic complications of the abnormal immune activation associated with HIV chronic disease.
Conclusions
HIV infection disrupts phenotypes and functions of monocytes, NK cells and ILCs, and subsequently the related adaptive host immune responses. ART restores some phenotypic and functional abnormalities associated with HIV infection, although persistent disruption of phenotypes and function of monocytes, NK cells, and innate lymphoid cells have been observed amongst populations of Art-treated adults. A further agreement of specific persistent innate immune prison cell phenotypic and functional abnormalities during Art is required to inform innovations in immune modulation interventions to optimize recovery of both innate and adaptive allowed organisation. Similarly, a further agreement of the drivers of persistent immune activation is required to inform strategic therapeutic interventions to minimize its complications, particularly in sub-Saharan Africa where other infectious causes of immune activation such as malaria, tuberculosis and helminthic infections are still endemic.
References
-
Medzhitov R, Janeway C Jr. Innate amnesty. North Engl J Med. 2000;343(five):338–44.
-
Chaplin DD. Overview of the immune response. J Allergy Clin Immunol. 2010;125(two):S3–23.
-
Ferreira C, et al. Differential survival of naive CD4 and CD8 T cells. J Immunol. 2000;165(7):3689–94.
-
Rivera A, et al. Innate cell communication boot-starts pathogen-specific immunity. Nat Immunol. 2016;17(4):356–63.
-
Iwasaki A, Medzhitov R. Command of adaptive amnesty by the innate immune organization. Nat Immunol. 2015;16(four):343.
-
Yona Due south, Jung S. Monocytes: subsets, origins, fates and functions. Curr Opin Hematol. 2010;17(i):53–9.
-
Ziegler-Heitbrock L. The CD14+ CD16+ claret monocytes: their role in infection and inflammation. J Leukoc Biol. 2007;81(3):584–92.
-
Terunuma H, et al. Potential role of NK cells in the consecration of immune responses: implications for NK cell–based immunotherapy for cancers and viral infections. Int Rev Immunol. 2008;27(3):93–110.
-
Vivier E, Ugolini S. NK cells: receptors and functions. Nat Rev Immunol. 2010;2:18.
-
Leavy O. Innate-like lymphocytes: volition the real ILC1 please stand up? Nat Rev Immunol. 2013;xiii(2):67.
-
Hepworth MR, et al. Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature. 2013;498(7452):113.
-
Douek DC, et al. HIV preferentially infects HIV-specific CD4+ T cells. Nature. 2002;417(6884):95.
-
Appay V, Kelleher AD. Immune activation and immune aging in HIV infection. Curr Opin HIV AIDS. 2016;11(2):242–ix.
-
Paiardini M, Müller-Trutwin Yard. HIV-associated chronic immune activation. Immunol Rev. 2013;254(1):78–101.
-
Sandler NG, Sereti I. Can early therapy reduce inflammation? Curr Opin HIV AIDS. 2014;9(i):72–nine.
-
Schuetz A, et al. Early ART initiation prevents disruption of the mucosal bulwark and subsequent T-cell activation. CROI Abstruse. 2014.
-
Amirayan-Chevillard Due north, et al. Impact of highly active anti-retroviral therapy (HAART) on cytokine product and monocyte subsets in HIV-infected patients. Clin Exp Immunol. 2000;120(i):107–12.
-
Funderburg NT, et al. Dynamics of immune reconstitution and activation markers in HIV+ treatment-naive patients treated with raltegravir, tenofovir disoproxil fumarate and emtricitabine. PLoS ONE. 2013;8(12):e83514.
-
Lederman MM. Immune restoration and CD4+ T-cell function with antiretroviral therapies. Aids. 2001;fifteen:S11–five.
-
Nabatanzi R, et al. Low antigen-specific CD4 T-cell immune responses despite normal absolute CD4 counts later long-term antiretroviral therapy an African accomplice. Immunol Lett. 2014;162(two):264–72.
-
Nakanjako D, et al. Dumb T-cell proliferation among HAART-treated adults with suboptimal CD4 recovery in an African cohort. BMC Immunol. 2013;14(i):26.
-
Nakanjako D, et al. Loftier T-cell immune activation and immune exhaustion among individuals with suboptimal CD4 recovery after 4 years of antiretroviral therapy in an African cohort. BMC Infect Dis. 2011;11(1):43.
-
Ancuta P, et al. Microbial translocation is associated with increased monocyte activation and dementia in AIDS patients. PLoS I. 2008;3(6):e2516.
-
Klatt NR, Funderburg NT, Brenchley JM. Microbial translocation, immune activation, and HIV illness. Trends Microbiol. 2013;21(1):6–13.
-
Kim WK, et al. Monocyte heterogeneity underlying phenotypic changes in monocytes according to SIV disease stage. J Leukoc Biol. 2010;87(4):557–67.
-
Sassé T, et al. Monocytes and their role in human immunodeficiency virus pathogenesis. Am J Infect Dis. 2012;8(ii):92.
-
Anzinger JJ, et al. Monocytes as regulators of inflammation and HIV-related comorbidities during cART. J Immunol Res. 2014. https://doi.org/ten.1155/2014/569819.
-
Chen P, et al. Perturbations of monocyte subsets and their association with T helper cell differentiation in acute and chronic HIV-1-infected patients. Front Immunol. 2017;8:272.
-
Jansen CA, et al. Analysis of the effect of highly agile antiretroviral therapy during acute HIV-1 infection on HIV-specific CD4 T prison cell functions. Aids. 2005;19(11):1145–54.
-
Le T, et al. Enhanced CD4+ T-cell recovery with earlier HIV-1 antiretroviral therapy. N Engl J Med. 2013;368(3):218–30.
-
Okoye AA, Picker LJ. CD4+ T-prison cell depletion in HIV infection: mechanisms of immunological failure. Immunol Rev. 2013;254(1):54–64.
-
Poovan Thousand, Ipp H, Glashoff RH. Monocytes in chronic HIV-1 infection: putative gut homing markers and their human relationship with allowed activation. AIDS Res Hum Retroviruses. 2014;30(S1):A128.
-
Tippett Due east, et al. Differential expression of CD163 on monocyte subsets in healthy and HIV-ane infected individuals. PLoS ONE. 2011;vi(5):e19968.
-
Han J, et al. CD14highCD16+ rather than CD14lowCD16+ monocytes correlate with disease progression in chronic HIV-infected patients. JAIDS J Acquir Immune Defic Syndr. 2009;52(5):553–9.
-
Burdo Th, Lackner A, Williams KC. Monocyte/macrophages and their part in HIV neuropathogenesis. Immunol Rev. 2013;254(1):102–13.
-
Marchetti One thousand, Tincati C, Silvestri 1000. Microbial translocation in the pathogenesis of HIV infection and AIDS. Clin Microbiol Rev. 2013;26(1):2–18.
-
Strauss-Ayali D, Conrad SM, Mosser DM. Monocyte subpopulations and their differentiation patterns during infection. J Leukoc Biol. 2007;82(2):244–52.
-
Michailidis C, et al. Impaired phagocytosis amidst patients infected past the human immunodeficiency virus: implication for a office of highly active anti-retroviral therapy. Clin Exp Immunol. 2012;167(three):499–504.
-
De Maria A, et al. The impaired NK cell cytolytic role in viremic HIV-1 infection is associated with a reduced surface expression of natural cytotoxicity receptors (NKp46, NKp30 and NKp44). Eur J Immunol. 2003;33(9):2410–8.
-
Mavilio D, et al. Natural killer cells in HIV-1 infection: dichotomous effects of viremia on inhibitory and activating receptors and their functional correlates. Proc Natl Acad Sci. 2003;100(25):15011–6.
-
Schafer JL, et al. Accumulation of cytotoxic CD16+ NK cells in simian immunodeficiency virus-infected lymph nodes associated with in situ differentiation and functional anergy. J Virol. 2015;89(thirteen):6887–94.
-
Iannello A, et al. Antiviral NK prison cell responses in HIV infection: I. NK jail cell receptor genes as determinants of HIV resistance and progression to AIDS. J Leukoc Biol. 2008;84(1):one–26.
-
Alter G, et al. Sequential deregulation of NK cell subset distribution and function starting in acute HIV-1 infection. Blood. 2005;106(x):3366–9.
-
Alter G, Altfeld M. NK cells in HIV-1 infection: evidence for their role in the control of HIV-1 infection. J Intern Med. 2009;265(1):29–42.
-
Junior T, Nascimento O, et al. Study of natural cytotoxicity receptors in patients with HIV/AIDS and cancer: a cross-sectional study. Sci World J. 2016. https://doi.org/10.1155/2016/2085871.
-
Bayigga L, et al. High CD56++ CD16-natural killer (NK) cells among suboptimal immune responders later four years of suppressive antiretroviral therapy in an African adult HIV treatment cohort. BMC Immunol. 2014;15(1):1.
-
De Maria A, Moretta L. NK cell function in HIV-i infection. Curr HIV Res. 2008;6(5):433–forty.
-
Jensen SS, et al. Restoration of the NK cells ability to mediate ADCC in HIV-1 positives afterwards six months of HAART can be explained past normalization of their phenotype. AIDS Res Hum Retroviruses. 2014;30(S1):A126.
-
Naluyima P, et al. Impaired natural killer cell responses are associated with loss of the highly activated NKG2A+ CD57+ CD56dim subset in HIV-1 subtype D infection in Uganda. AIDS (London, England). 2014;28(9):1273.
-
Sips M, et al. Altered distribution of mucosal NK cells during HIV infection. Mucosal Immunol. 2012;5(i):30–40.
-
Altfeld M, et al. DCs and NK cells: critical effectors in the allowed response to HIV-i. Nat Rev Immunol. 2011;xi(3):176–86.
-
Azzoni L, et al. Sustained impairment of IFN-γ secretion in suppressed HIV-infected patients despite mature NK cell recovery: evidence for a defective reconstitution of innate immunity. J Immunol. 2002;168(xi):5764–70.
-
Chehimi J, et al. Baseline viral load and allowed activation determine the extent of reconstitution of innate immune effectors in HIV-i-infected subjects undergoing antiretroviral treatment. J Immunol. 2007;179(4):2642–50.
-
Frias M, et al. Persistence of pathological distribution of NK cells in HIV-infected patients with prolonged utilize of HAART and a sustained immune response. PLoS 1. 2015;10(3):e0121019.
-
Luo Z, et al. Increased natural killer jail cell activation in HIV-infected immunologic non-responders correlates with CD4+ T cell recovery after antiretroviral therapy and viral suppression. PLoS ONE. 2017;12(ane):e0167640.
-
Parato KG, et al. Normalization of natural killer cell part and phenotype with effective anti-HIV therapy and the office of IL-ten. Aids. 2002;sixteen(ix):1251–vi.
-
Bayigga L, et al. High CD56++ CD16-natural killer (NK) cells among suboptimal allowed responders afterward four years of suppressive antiretroviral therapy in an African adult HIV handling cohort. BMC Immunol. 2014;15(one):2.
-
Flynn AG, et al. Socioeconomic position and x-year survival and virologic outcomes in a Ugandan HIV accomplice receiving antiretroviral therapy. PLoS ONE. 2017;12(12):e0189055.
-
Walker JA, Barlow JL, McKenzie AN. Innate lymphoid cells—how did we miss them? Nat Rev Immunol. 2013;13(two):75–87.
-
Almeida F, Belz M. Innate lymphoid cells: models of plasticity for allowed homeostasis and rapid responsiveness in protection. Mucosal Immunol. 2016;9(5):1103–12.
-
Brenchley JM, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med. 2006;12(12):1365–71.
-
Li H, et al. Hypercytotoxicity and rapid loss of NKp44+ innate lymphoid cells during acute SIV infection. PLoS Pathog. 2014;10(12):e1004551.
-
Reeves RK, et al. Depletion of lamina propria innate lymphoid cells in simian immunodeficiency virus infection. AIDS Res Hum Retroviruses. 2014;thirty(12):1160–1.
-
Xu H, et al. IL-17-producing innate lymphoid cells are restricted to mucosal tissues and are depleted in SIV-infected macaques. Mucosal Immunol. 2012;v(6):658–69.
-
Kløverpris HN, et al. Innate lymphoid cells are depleted in HIV infection. AIDS Res Hum Retroviruses. 2014;30(S1):A14.
-
Kløverpris HN, et al. Innate lymphoid cells are depleted irreversibly during acute HIV-1 infection in the absence of viral suppression. Immunity. 2016;44(2):391–405.
-
Mudd JC, Brenchley JM. ILC you after: early and irreparable loss of innate lymphocytes in HIV infection. Immunity. 2016;44(2):216–viii.
-
McKenzie AN, Spits H, Eberl G. Innate lymphoid cells in inflammation and immunity. Immunity. 2014;41(3):366–74.
-
Krämer B, et al. Compartment-specific distribution of homo abdominal innate lymphoid cells is altered in HIV patients under constructive therapy. PLoS Pathog. 2017;13(5):e1006373.
-
Shelburne SA, et al. Immune reconstitution inflammatory syndrome: emergence of a unique syndrome during highly active antiretroviral therapy. Medicine. 2002;81(iii):213–27.
-
Meya DB, et al. The immunopathogenesis of cryptococcal immune reconstitution inflammatory syndrome: understanding a puzzler. Curr Opin Infect Dis. 2016;29(1):10–22.
-
Wilson EM, Sereti I. Immune restoration afterward antiretroviral therapy: the pitfalls of jerky or incomplete repairs. Immunol Rev. 2013;254(1):343–54.
-
Tran HTT, et al. The role of monocytes in the development of tuberculosis-associated immune reconstitution inflammatory syndrome. Immunobiology. 2014;219(1):37–44.
-
Tran HT, et al. Modulation of the complement system in monocytes contributes to tuberculosis-associated immune reconstitution inflammatory syndrome. Aids. 2013;27(11):1725–34.
-
Andrade BB, et al. Mycobacterial antigen driven activation of CD14++ CD16− monocytes is a predictor of tuberculosis-associated immune reconstitution inflammatory syndrome. PLoS Pathog. 2014;10(ten):e1004433.
-
Pean P, et al. Natural killer prison cell degranulation capacity predicts early onset of the allowed reconstitution inflammatory syndrome (IRIS) in HIV-infected patients with tuberculosis. Blood. 2012;119(xiv):3315–twenty.
-
Conradie F, et al. Natural Killer cell activation distinguishes M. tuberculosis-mediated immune reconstitution syndrome (IRIS) from chronic HIV and HIV-MTB co-infection. J Acquir Allowed Defic Syndr (1999). 2011;58(3):309.
-
d'Ettorre G, et al. HIV-associated immune activation: from demote to bedside. AIDS Res Hum Retroviruses. 2011;27(four):355–64.
-
Maidji E, et al. Replication of CMV in the gut of HIV-infected individuals and epithelial bulwark dysfunction. PLoS Pathog. 2017;13(ii):e1006202.
-
Appay Five, Sauce D. Allowed activation and inflammation in HIV-i infection: causes and consequences. J Pathol. 2008;214(2):231–41.
-
Sun SC. Not-approved NF-κB signaling pathway. Prison cell Res. 2011;21(1):71–85.
-
Kawakami 1000, Scheidereit C, Roeder RG. Identification and purification of a homo immunoglobulin-enhancer-bounden poly peptide (NF-kappa B) that activates transcription from a human immunodeficiency virus type i promoter in vitro. Proc Natl Acad Sci. 1988;85(13):4700–iv.
-
Appay Five, Sauce D. Assessing immune crumbling in HIV-infected patients. Virulence. 2016;eight:one–ten.
-
Crowe SM, et al. The macrophage: the intersection betwixt HIV infection and atherosclerosis. J Leukoc Biol. 2010;87(four):589–98.
-
Chase Pw, Lee SA, Siedner MJ. Immunologic biomarkers, morbidity, and mortality in treated HIV infection. J Infect Dis. 2016;214(Suppl 2):S44–50.
-
Duprez DA, et al. Lipoprotein particle subclasses, cardiovascular disease and HIV infection. Atherosclerosis. 2009;207(2):524–9.
-
Ssinabulya I, et al. Subclinical atherosclerosis among HIV-infected adults attending HIV/AIDS intendance at two big ambulatory HIV clinics in Uganda. PLoS I. 2014;nine(2):e89537.
-
Nakanjako D, et al. CD4 T-cell activation and reduced regulatory T-cell populations are associated with early development of cataracts amongst HIV-infected adults in Uganda. Immunol Lett. 2014;161(1):44–9.
-
Shiels MS, Pfeiffer RM, Engels EA. Historic period at cancer diagnosis amid persons with AIDS in the United States. Ann Intern Med. 2010;153(7):452–sixty.
-
Silverberg MJ, Chao C, Abrams DI. New insights into the role of HIV infection on cancer risk. Lancet Oncol. 2009;10(12):1133–4.
-
Arnsten JH, et al. Decreased os mineral density and increased fracture take a chance in aging men with or at take a chance for HIV infection. AIDS. 2007;21(5):617–23.
-
Choi AI, et al. HIV-infected persons continue to lose kidney function despite successful antiretroviral therapy. AIDS. 2009;23(16):2143–ix.
-
McCutchan JA, et al. HIV suppression past HAART preserves cerebral function in advanced, immune-reconstituted AIDS patients. AIDS. 2007;21(9):1109–17.
Authors' contributions
RN, SC, MJ, SRJ, DN contributed to the conceptualization and execution of the review. RN and DN drafted the manuscript. All authors reviewed manuscript for publication. All authors read and canonical the final manuscript.
Acknowledgements
The authors also acknowledge funding from the Alliance for Global Health and Science at the University of California, Berkeley, U.s.a. and the Wheeler Center for Emerging and Neglected Diseases, University of California, Berkeley, USA that funded our inquiry to further sympathise Natural Killer cells and Innate allowed cells during Art.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
Non applicable.
Consent for publication
Not applicable.
Ethics approving and consent to participate
Not applicable.
Funding
This work was supported through the DELTAS Africa Initiative [Grant# 107743/Z/15/Z], that funded Damalie Nakanjako and Rose Nabatanzi through a group leader award. The DELTAS Africa Initiative is an independent funding scheme of the African Academy of Sciences (AAS)'due south Alliance for Accelerating Excellence in Science in Africa (AESA) and supported by the New Partnership for Africa's Evolution Planning and Coordinating Agency (NEPAD Agency) with funding from the Wellcome Trust [Grant #107743/Z/15/Z] and the UK authorities. The views expressed in this publication are those of the author(southward) and not necessarily those of AAS, NEPAD Agency, Wellcome Trust or the UK government.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Affiliations
Corresponding author
Rights and permissions
Open Access This commodity is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you lot requite appropriate credit to the original author(s) and the source, provide a link to the Creative Eatables license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zilch/1.0/) applies to the information made available in this article, unless otherwise stated.
Reprints and Permissions
About this article
Cite this article
Nabatanzi, R., Cose, Due south., Joloba, Thou. et al. Furnishings of HIV infection and Art on phenotype and part of circulating monocytes, natural killer, and innate lymphoid cells. AIDS Res Ther 15, 7 (2018). https://doi.org/ten.1186/s12981-018-0194-y
-
Received:
-
Accustomed:
-
Published:
-
DOI : https://doi.org/x.1186/s12981-018-0194-y
Keywords
- HIV
- Innate immunity
- Monocytes
- Natural killer cells
- Innate lymphoid cells
- Antiretroviral therapy
Source: https://aidsrestherapy.biomedcentral.com/articles/10.1186/s12981-018-0194-y
0 Response to "Clip Art of How Aids Affects Your T Cells"
Enviar um comentário